Research from Dr. Xueqing Zhang’s group at Shanghai Jiao Tong University on steroid structured inhalable LNPs for co delivery of steroid drugs and anti TSLP mRNA for synergistic treatment of refractory asthma was published in Nature Communications
Severe asthma is a complex and difficult-to-treat inflammatory disease, and its clinical management faces significant challenges, particularly as some patients develop resistance to glucocorticoid (GC) therapy, resulting in poor disease control and recurrent acute exacerbations that severely affect patient prognosis. Studies have shown that airway epithelial injury can induce aberrantly high expression of thymic stromal lymphopoietin (TSLP), which promotes phosphorylation of the glucocorticoid receptor (GR) by activating the p38 MAPK and STAT3 signaling axes, thereby inhibiting its nuclear translocation and ultimately weakening the anti-inflammatory effects of GCs, constituting one of the key mechanisms of GC resistance. Although antibody-based therapies such as anti-TSLP monoclonal antibodies have entered clinical application, risks of systemic drug accumulation caused by off-target effects, as well as insufficient local drug exposure in lung tissues, still limit their sustained inhibitory efficacy on TSLP signaling within the local inflammatory microenvironment. Therefore, there is an urgent need to develop efficient and targeted novel therapeutic strategies to overcome GC resistance in severe asthma.
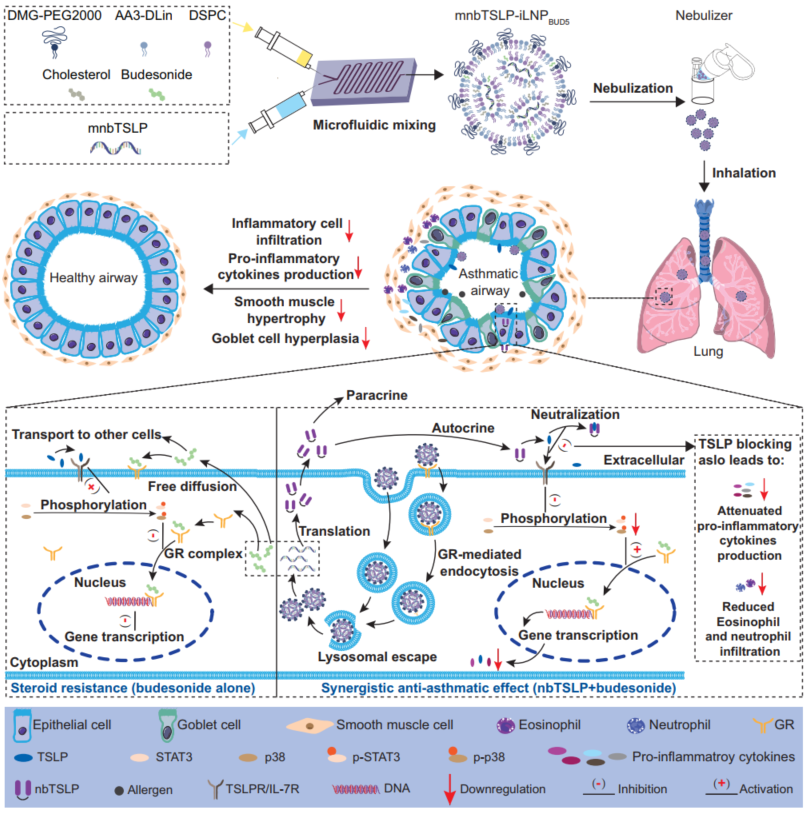
Recently, the team led by Professor Zhang Xueqing at the School of Pharmacy, Shanghai Jiao Tong University, in collaboration with Professor Xu Xiaoyang’s team at the New Jersey Institute of Technology, published a research article in Nature Communications entitled “Budesonide-incorporated inhalable lipid nanoparticles for anti-TSLP nanobody mRNA delivery to treat steroid-resistant asthma.” This study innovatively constructed a novel steroid-structured inhalable LNP delivery system (ASCEND), which, through a hydrophobic chimeric design integrating steroid molecules such as budesonide with the lipid scaffold, enables lung-targeted inhalation delivery of mRNA encoding an anti-TSLP nanobody (mnbTSLP). This strategy aims to achieve targeted accumulation and synergistic action of steroid anti-inflammatory drugs and antibody therapeutics in the lungs, by modulating the airway inflammatory microenvironment and blocking TSLP-mediated type 2 inflammatory pathways, thereby providing an innovative therapeutic approach for severe refractory asthma with both rapid onset and immunomodulatory advantages.

In this project, the researchers used budesonide as a representative anti-asthmatic steroid drug. Based on its structural similarity to the cholesterol component in LNPs, they proposed partially replacing cholesterol with budesonide as a structural lipid component. Combined with formulation optimization, they successfully developed a steroid-structured inhalable LNP (iLNP BUD5). After nebulization, this iLNP maintained stable particle size and morphology without loss of mRNA encapsulation efficiency, and the aerosol efficiently mediated local transfection, expression, and secretion of mnbTSLP in the lungs. Animal studies demonstrated that, following nebulized administration, mnbTSLP-iLNP BUD5 exhibited significant accumulation in the lungs and achieved efficient cellular uptake and lysosomal escape via a GR-mediated endocytic pathway. The released mnbTSLP was translated in the cytoplasm, and the produced and secreted nbTSLP nanobody specifically bound to and neutralized excessive extracellular TSLP, thereby blocking downstream signaling activation. Meanwhile, the structurally integrated budesonide synergistically exerted its classical anti-inflammatory effects. This ASCEND strategy innovatively integrates small-molecule drugs with gene therapy, providing a new therapeutic paradigm for addressing glucocorticoid-resistant severe asthma. This delivery platform has the following three key highlights: (1) through multidimensional synergistic optimization of formulation components, preparation processes, and nebulization systems, and on this basis integrating anti-asthmatic steroid drugs as structural components, a novel inhalable mRNA-LNP system was constructed to achieve precise pulmonary delivery of both small-molecule and nucleic acid drugs; (2) synergistic anti-inflammatory effects: leveraging the synergistic effects between the carrier and nucleic acid therapeutics, airway inflammation and the TSLP signaling pathway were significantly suppressed, and the combination therapy markedly alleviated airway inflammation, remodeling, and hyperresponsiveness in mouse models, while mnbTSLP restored sensitivity of asthmatic mice to budesonide by inhibiting key inflammatory pathways; (3) high patient compliance: this strategy employs noninvasive inhalation administration, which is simple to operate and highly acceptable to patients, with broad clinical application prospects.
Schematic illustration of inhaled mscFv/siPD-L1@LNP inducing antitumor immunity in the lung.
Based on the novel inhalable lung-targeted delivery platform ASCEND, the carrier and the loaded mRNA synergistically achieve a mechanistic breakthrough in the treatment of refractory asthma. The research team will further optimize the delivery system to promote the clinical translation of ASCEND therapy, providing safer and more effective treatment options for patients with severe asthma. In addition, the inhalable mRNA–steroid combination strategy pioneered in this study can be extended to the treatment of respiratory diseases such as chronic obstructive pulmonary disease (COPD) and pulmonary fibrosis, offering a new delivery system to support clinical translation in respiratory medicine.
Original article link:
Original article link: https://doi.org/10.1038/s41467-025-61114-4
