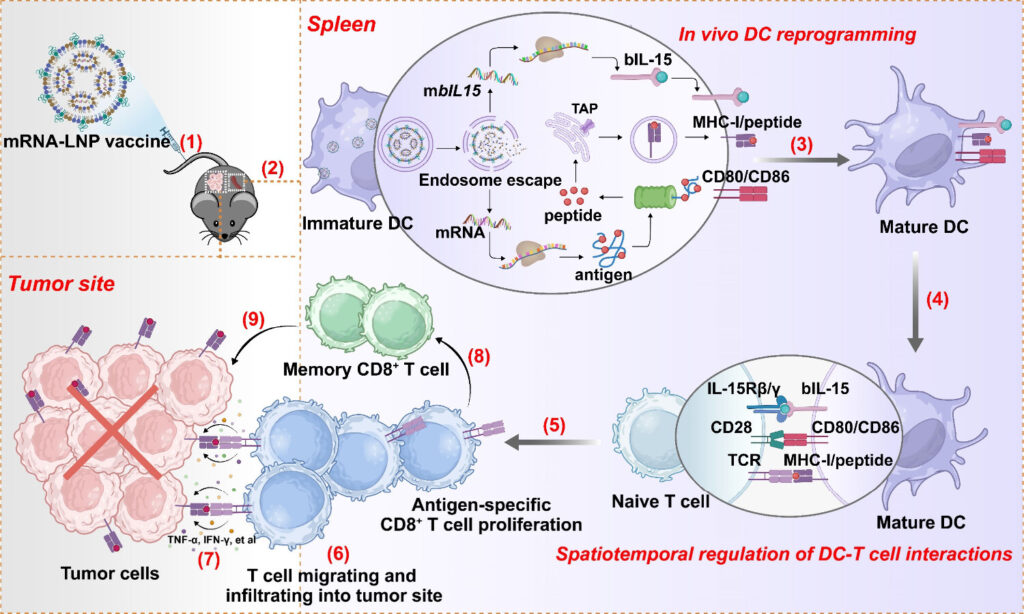
Research from Dr. Xueqing Zhang’s group at Shanghai Jiao Tong University on mRNA vaccine mediated in vivo dendritic cell reprogramming to elicit potent antitumor immune responses was published in ACS Nano

The spleen, as the largest secondary lymphoid organ in the body, is enriched with DCs and T cells and represents an ideal physiological site for initiating T cell responses via MHC-I-mediated antigen presentation on DCs. In this study, optimized lipid nanoparticles were employed to selectively deliver mRNA encoding tumor antigens and a membrane-bound IL-15/IL-15Rα complex (bIL-15, the biologically active form of IL-15) into splenic DCs. Following systemic administration, the VISIT platform successfully achieved in vivo DC reprogramming, enabling the simultaneous presentation of antigen/MHC-I complexes and bIL-15 on the DC surface. This “two-in-one” design allows DCs to trans-present both tumor antigens and bIL-15 to CD8⁺ T cells, thereby effectively promoting the in situ generation and expansion of antigen-specific CTLs, driving tumor clearance and immune memory formation, while minimizing the risk of nonspecific immune activation.