Research from Dr. Xueqing Zhang’s group at Shanghai Jiao Tong University on inhalable lung targeted mRNA delivery was published in Nature Communications
Idiopathic pulmonary fibrosis (IPF) is one of the most common interstitial lung diseases, characterized by extensive fibrotic lesions and a mortality rate higher than that of many cancers. Current therapeutic agents for IPF, including pirfenidone and nintedanib, can only slow disease progression but are unable to reverse established fibrotic scars. Inhalable mRNA-based protein replacement therapy enables maximal drug accumulation in the lungs, allowing for the expression of virtually any desired protein or peptide, targeting “undruggable” pathways, and generating therapeutic antibody cocktails. This approach holds great promise for the treatment of respiratory diseases, including IPF. However, successful inhalable nucleic acid delivery systems must withstand shear stress during nebulization, penetrate mucus barriers, evade macrophage clearance, and ensure efficient cytoplasmic delivery and protein translation.
Recently, the team published an article titled “Optimized inhaled LNP formulation for enhanced treatment of idiopathic pulmonary fibrosis via mRNA-mediated antibody therapy” in Nature Communications. In this study, the authors developed an inhalable nucleic acid nanocarrier capable of delivering mRNA to the lungs of fibrotic mice, thereby achieving antifibrotic therapeutic effects. Dr. Xin Bai from Shanghai Jiao Tong University is the first author, and the School of Pharmacy at Shanghai Jiao Tong University serves as both the primary and corresponding institution.

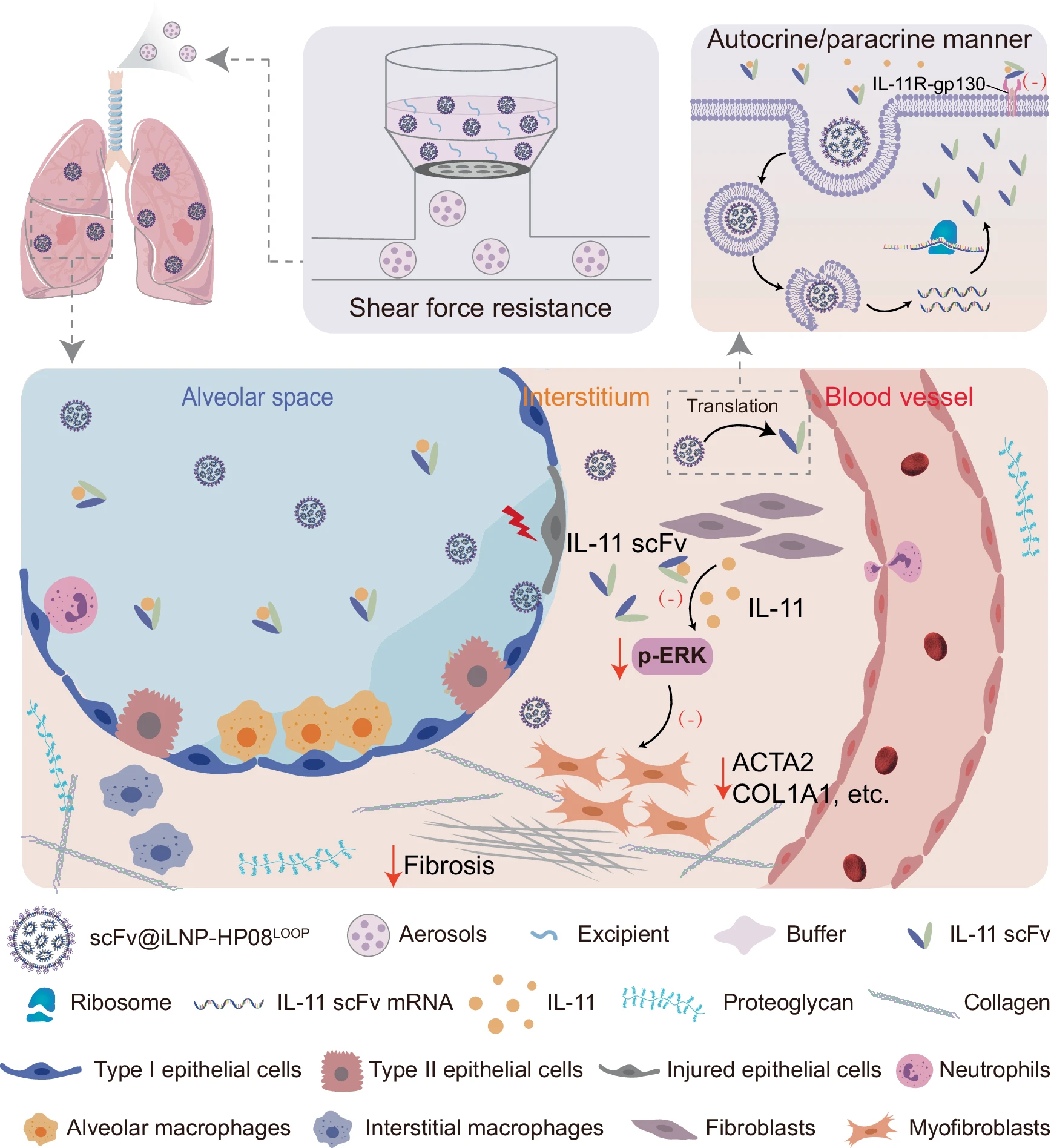
In this project, the researchers first proposed a novel “LOOP” platform, consisting of a four-step workflow that avoids extensive and redundant LNP screening. Using microfluidic technology, they prepared a series of LNPs composed of varying molar ratios of ionizable lipids, cholesterol, helper lipids, and PEG-lipids, from which formulations with optimal resistance to shear stress were selected. Subsequently, the dialysis system for these LNPs was optimized. Meanwhile, excipients such as alcohols and nonionic surfactants were introduced to enhance the resistance of LNPs to shear-induced damage during nebulization. Finally, based on the selected dialysis buffer and excipients, the molar ratios of lipid components were further tuned to investigate the structure–activity relationship between lipid composition and luciferase expression. Ultimately, an optimized formulation, iLNP-HP08LOOPcharacterized by a high proportion of helper lipids, an acidic dialysis buffer, and excipients—was identified, exhibiting superior nebulization stability and enhanced pulmonary luciferase expression.
吸入式肺靶向疗法治疗IPF的示意图
Based on this optimized iLNP-HP08LOOPdelivery system, the researchers further loaded mRNA encoding an IL-11 single-chain antibody (scFv) to generate scFv@iLNP-HP08LOOP, thereby successfully developing an mRNA-mediated inhalable antibody therapy. Following administration via a vibrating mesh nebulizer, scFv@iLNP-HP08LOOPefficiently deposited in the lungs, where IL-11 scFv mRNA was locally released and subsequently translated into IL-11 scFv protein. The expressed antibody binds to overexpressed IL-11 in the pulmonary microenvironment through autocrine or paracrine mechanisms. In a bleomycin-induced pulmonary fibrosis mouse model, inhaled scFv@iLNP-HP08LOOPeffectively inhibited fibroblast activation and extracellular matrix (ECM) production, demonstrating significantly superior efficacy compared to nebulized or intravenously administered IL-11 scFv protein. Pulmonary function tests further showed that inhaled scFv@iLNP-HP08LOOPsignificantly improved all lung function parameters in fibrotic mice. Notably, the HP08LOOPsystem also enhanced the nebulization stability and pulmonary luciferase expression of LNPs formulated with the clinically used ionizable lipid ALC0315.
In summary, the “LOOP” strategy provides a versatile platform for the development of other inhalable mRNA-based protein replacement therapies, offering broad prospects for the prevention and management of interstitial lung diseases (ILDs), including IPF, as well as other respiratory disorders.
Original article link:
https://www.nature.com/articles/s41467-024-51056-8
