Research from Dr. Xueqing Zhang’s group at Shanghai Jiao Tong University on computer aided design of ionizable lipids to overcome liver accumulation of LNPs was published in Journal of the American Chemical Society
As a new-generation biotechnology platform, mRNA therapeutics have demonstrated revolutionary application potential. Four-component lipid nanoparticles (LNPs), composed of ionizable lipids, cholesterol, phospholipids, and polyethylene glycol (PEG)-lipids, represent the current mainstream mRNA delivery platform. However, clinically used four-component LNP formulations, following systemic administration, predominantly accumulate in the liver due to apolipoprotein E/low-density lipoprotein receptor (ApoE/LDLR)-mediated hepatocyte uptake. This intrinsic liver tropism greatly limits the delivery efficiency of mRNA therapeutics to non-hepatic tissues and constrains their clinical application potential across a broader range of diseases.
Recently, the team led by Zhang Xueqing at the School of Pharmacy, Shanghai Jiao Tong University, in collaboration with Xu Xiaoyang’s group at the New Jersey Institute of Technology, published a research article in the Journal of the American Chemical Society entitled “Computationally Aided Design of Ionizable Cholesteryl Lipids for Lipid Nanoparticles to Modulate Hepatic mRNA Accumulation.” This study proposes a computationally assisted design strategy for ionizable lipids and develops and optimizes a class of sterol-derived ionizable lipids (iChol-lipids) with low ApoE binding capacity. Compared with cholesterol, iChol-lipids exhibit significantly reduced ApoE affinity and can self-assemble with helper lipids and PEG-lipids to form stable cholesterol-free three-component LNPs (Tc-LNPs), enabling efficient mRNA delivery while substantially attenuating ApoE/LDLR-mediated hepatic accumulation. In addition, this computational design strategy is also applicable to ALC-0315 (a lipid component used in approved vaccines), providing a new approach for accelerating the development of lipid nanocarriers with extrahepatic delivery capability and significantly expanding the clinical application scope of mRNA therapeutics. Teng Yilong (postdoctoral fellow) and Guo Yuxuan (master’s student) from the School of Pharmacy, Shanghai Jiao Tong University, are co-first authors of the paper, and the School of Pharmacy, Shanghai Jiao Tong University, serves as the primary and corresponding institution.

Conventional four-component LNPs rely on cholesterol to maintain structural stability; however, after systemic administration, such formulations exhibit pronounced liver accumulation, primarily because the high affinity between cholesterol and ApoE leads to substantial adsorption of ApoE onto the LNP surface, which in turn facilitates hepatocyte uptake via the ApoE/LDLR-mediated endocytic pathway. To overcome the liver tropism of traditional LNPs, this study employed DiffDock-L molecular docking technology to guide lipid molecular design. By integrating the cholesterol moiety, which stabilizes the LNP nanostructure, as a hydrophobic tail into the molecular scaffold of ionizable lipids, the researchers achieved chemical fusion of two core LNP components and successfully developed a novel iChol-lipid molecular system. Through a combinatorial chemistry strategy, a molecular library comprising 105 novel iChol-lipids was constructed. These iChol-lipids exhibit markedly reduced ApoE affinity compared with cholesterol and can self-assemble with helper lipids and lipid-PEG to form stable cholesterol-free three-component Tc-LNPs, enabling efficient RNA encapsulation while significantly weakening ApoE/LDLR-mediated hepatic accumulation of LNPs. Based on systematic in vitro screening, structure–activity relationship (SAR) analysis was performed on the iChol-lipid library, leading to the identification of 10 top-performing candidate compounds. Tc-LNPs assembled from these optimized iChol-lipids achieved efficient mRNA accumulation in extrahepatic tissues (such as spleen and muscle) while significantly reducing liver accumulation. Notably, Tc-LNPs constructed from two sterol-derived ionizable lipids, iChol15-C4A2 and iChol15-C4ACM, exhibited spleen-to-liver luciferase luminescence ratios of 4.78 and 5.20, respectively, which are substantially higher than those of conventional LNP formulations MC3 (0.24) and ALC-0315 (0.12). Further studies showed that, compared with traditional four-component LNPs, Tc-LNPs reduced ApoE adsorption by 90%, significantly suppressing ApoE/LDLR-mediated hepatic accumulation of mRNA while effectively enhancing delivery efficiency to extrahepatic organs such as the spleen. Moreover, this computationally assisted lipid design strategy is also applicable to the derivatization of commercial lipids (such as ALC-0315). By replacing the 2-hexyldecanoate tail with a cholesterol structure, the researchers synthesized two cholesterol-functionalized ALC-0315 derivatives. Compared with ALC-0315 LNPs, which exhibit pronounced liver tropism, the three-component Tc-LNPs prepared from these derivatives showed significantly improved mRNA delivery efficiency to the spleen.
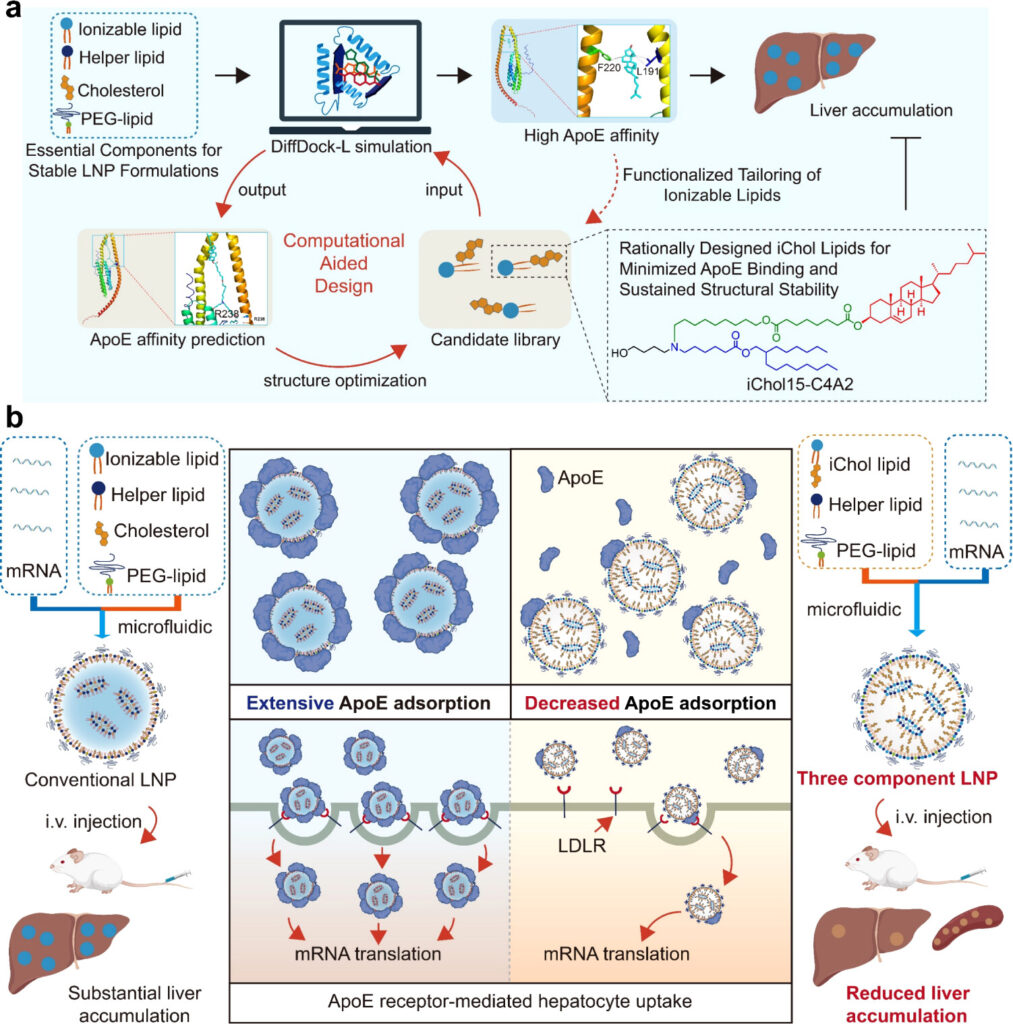
This study establishes an efficient ionizable lipid design platform that, by strategically weakening the interaction between lipid components and ApoE, significantly reduces the liver accumulation tendency of LNPs and achieves efficient mRNA delivery to extrahepatic tissues such as the spleen. Proof-of-concept experiments further validated the delivery performance of the lipid materials developed using this platform. These findings provide a feasible and innovative strategy for exploring the potential application of mRNA therapeutics in the treatment of extrahepatic diseases and open new avenues for expanding the therapeutic landscape of mRNA technologies.
Original article link: https://doi.org/10.1021/jacs.5c14870
