Collaborative research between Dr. Xueqing Zhang’s group at Shanghai Jiao Tong University and Dr. Xiaoyang Xu’s group at New Jersey Institute of Technology on mRNA LNP mediated pyroptosis therapy to enhance cancer immunotherapy was published in Nature Communications
Tumor immunotherapy, particularly immune checkpoint blockade (ICB) therapy, has become an important cancer treatment modality and has prolonged the overall survival of many cancer patients. However, only a small proportion of patients (10%–30% of those with solid tumors) achieve a complete response to immunotherapy, partly due to the highly immunosuppressive tumor microenvironment (TME) present in immunologically “cold” tumors. In tumor immunotherapy, the synergistic induction of T cell immune responses with inflammatory cytokines or immune agonists represents a promising strategy to alleviate immunosuppression and activate T cells. However, effective antitumor immunity requires the activation of all steps in the cancer immunity cycle, including immunogenic cell death (ICD), maturation of antigen-presenting cells, priming and activation of T cells, recruitment of tumor-infiltrating immune cells, and production of inflammatory cytokines. Unfortunately, even combination therapies using multiple agents often fail to achieve effective inhibition of cancer cells. Therefore, new therapeutic strategies are needed to induce effective antitumor immunity and broaden the scope of immunotherapy.
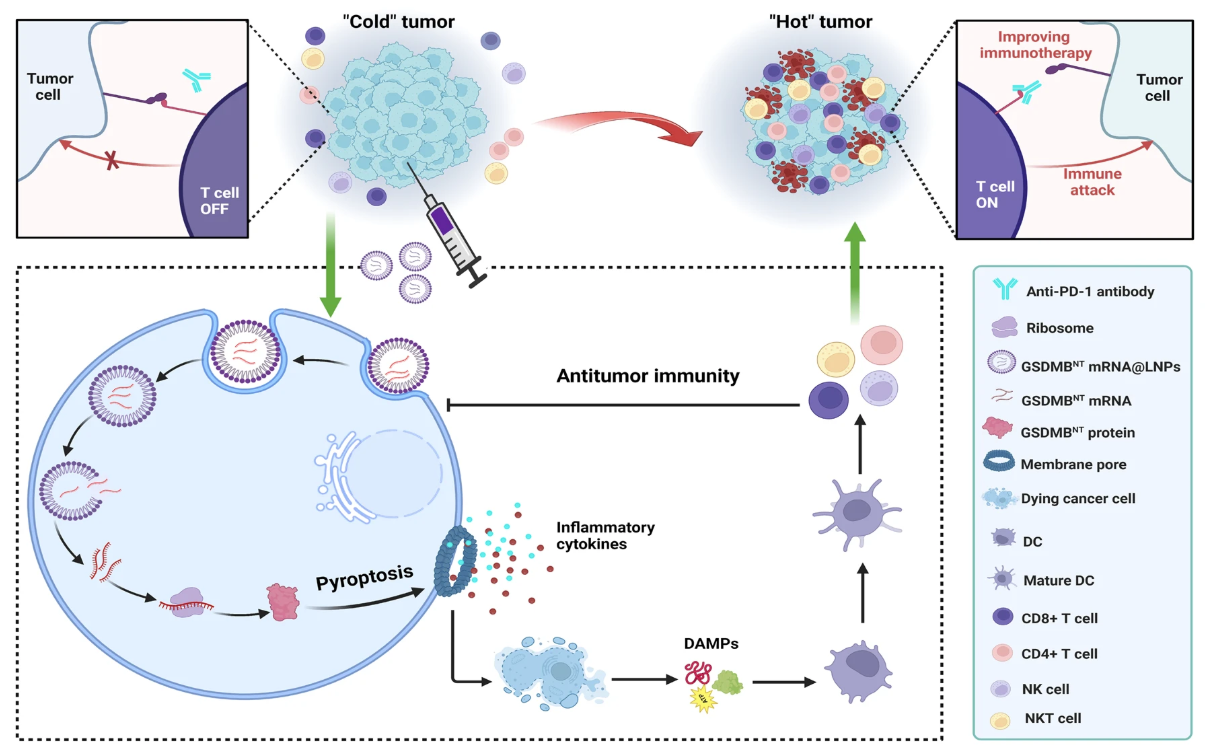
Recently, the team led by Professor Xueqing Zhang at Shanghai Jiao Tong University and Professor Xiaoyang Xu at the New Jersey Institute of Technology published a research article in Nature Communications entitled “mRNA lipid nanoparticle-mediated pyroptosis sensitizes immunologically cold tumors to checkpoint immunotherapy.” This study proposes an mRNA-based pyroptosis nanotherapeutic strategy capable of initiating the cancer immunity cycle and converting cold tumors into “hot” tumors characterized by inflammatory cytokine expression and T cell infiltration, thereby effectively treating immunologically cold tumors.

Pyroptosis is an inflammatory form of programmed cell death triggered by the proteolytic cleavage of members of the gasdermin (GSDM) family. GSDMs are typically autoinhibited through intramolecular interactions between their N-terminal and C-terminal domains. Upon cleavage of the linker region by specific cysteine-aspartic proteases and other proteases, the necrotic N-terminal domain oligomerizes and translocates to the plasma membrane. The liberated N-terminal domain binds to lipid components and forms pores in the cell membrane, leading to rapid plasma membrane rupture and the release of danger-associated molecular patterns (DAMPs) and proinflammatory cytokines. Immune cells recognize specific DAMPs, thereby triggering a cascade of immune responses, including immune cell activation and infiltration. In addition, proinflammatory cytokines released through pyroptotic pores help reverse the immunosuppressive tumor microenvironment (TME).Despite these encouraging findings, low expression of GSDMs in many cancers and the complexity of the cleavage process hinder the induction of pyroptosis via protease delivery to generate antitumor immunity.The research team hypothesized that directly delivering the N-terminal domain of GSDM to induce pyroptosis represents an effective approach to trigger a cascade of events in the cancer immunity cycle and convert cold tumors into hot tumors.Gene therapy based on mRNA nanomedicine holds broad clinical application prospects. Recently, a synthetically developed ionizable cationic lipid nanoparticle formulation (AA3-Dlin LNPs) has demonstrated favorable safety and high mRNA translation efficiency both in vitro and in vivo. mRNA-LNPs that cooperate with GSDM to activate pyroptosis are expected to play an important role in tumor therapy.
In this study, we proposed an mRNA-based nanomedicine approach in which the AA3-Dlin LNP formulation encapsulates a single mRNA encoding the N-terminal domain of GSDMBGSMDBNT mRNA@LNP. The developedGSMDBNT mRNA@LNP formulation is self-assembled from an ionizable cationic lipid (AA3-Dlin), phospholipids, cholesterol, and PEG, withGSMDBNT mRNA encapsulated within the LNP via electrostatic interactions.The research team envisioned that this LNP could be delivered to tumor tissues, where the mRNA would be translated into the N-terminal domain of the GSDMB protein, directly triggering pyroptosis without the need for proteolytic cleavage.Pyroptosis has the capacity to induce tumor cell death, initiate the release of proinflammatory cytokines, and activate and recruit intratumoral immune cells, thereby triggering a cascade of events that further promote cell death, cytokine release, and immune response activation. The resulting positive feedback loop can establish a favorable immunogenic “hot” tumor microenvironment, rendering cancer cells more sensitive to immune checkpoint blockade (ICB)-mediated immunotherapy and exhibiting superior tumor inhibition compared to monotherapy.
Original article link:
https://www.nature.com/articles/s41467-023-39938-9
